
All of these elements are classified as reactive nonmetals, which means that they are highly reactive. Fluorine, chlorine, bromine, iodine, and astatine are some of the halogen elements. They are all elements that can be found in the group 17 of the periodic table, which is composed of halogens. Fluorine gas interacts violently with a wide range of other elements and compounds, and it is often regarded as one of the most toxic chemicals known to science. As a free element, it is not found in the natural world. What is the most reactive nonmetal, and what is the reason behind this?īecause the noble gases constitute a distinct category distinguished by their lack of reactivity, the element fluorine is the nonmetal with the highest level of reactivity. The alkaline Earth metals are grouped together in Group They are quite reactive, however not as much as the alkali metals. 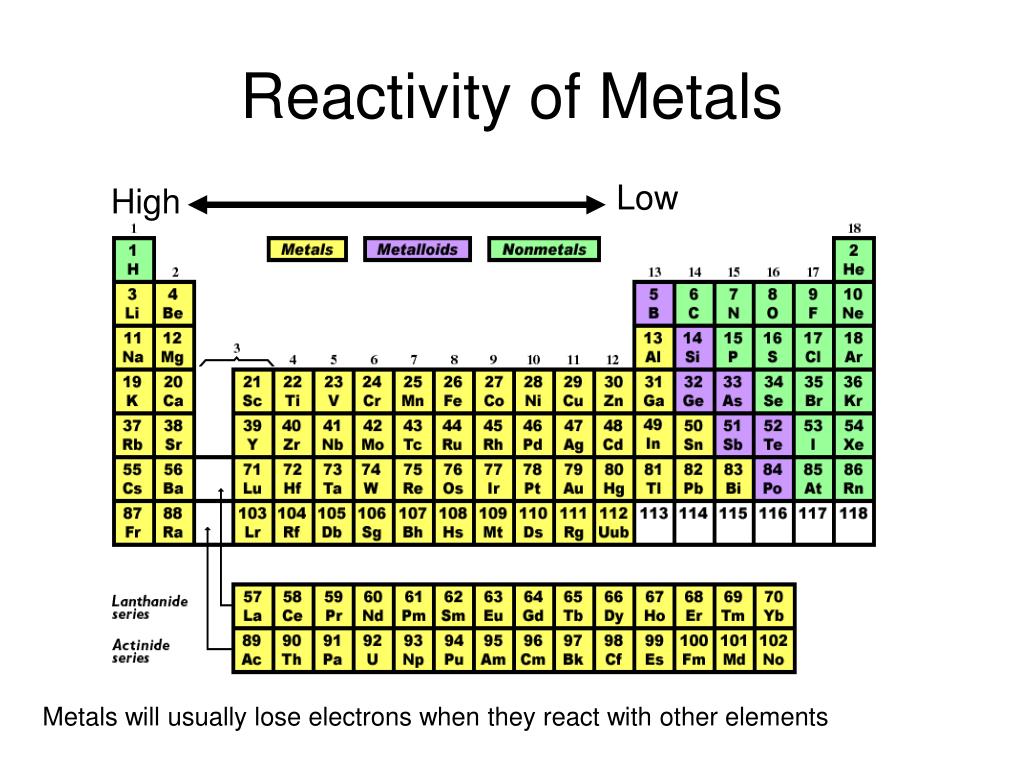
The alkali metals are the metals that are the most reactive. What are the names of the metals that are the most reactive? Make use of a mnemonic method to assist you in remembering the sequence of the components. Color may be used to aid in the learning of element groupings. 
Make up gibberish words out of the element symbols you have. Learn how to identify the different components in a song. Disperse the memorising process across a period of time. Techniques for Memorization Divide the table into parts for easier reading. How do I learn the elements in the periodic table? Which of the following halogens is the most reactive? A halogen, which is found in Group 17 of the periodic table, fluorine is the most reactive of all the elements, including chlorine and bromine. Which nonmetals have the highest reactivity?Īnswer and justification are as follows: Fluorine is the nonmetal with the highest reactivity. On the periodic table, this is the second most reactive family of elements, after the lanthanide family. Group II contains the alkaline earth metals, which are located just next door. What is the metal that is the second most reactive?Īs a result, we have completed our discussion of alkali metals in Group I. Which metal has the highest level of reactivity? There were 39 related questions and answers found. When it comes to alkali metals, which one is the most reactive? This is owing to the fact that the outer electron adds strength to the link between the atom’s nucleus and the outer electron. Due to its position on the periodic table, Francium should theoretically be the most reactive element however, due to the fact that it is also radioactive, Cesium takes that title instead.Īs a result, one would wonder why francium isn’t considered to be the most reactive metal.īecause the outer electron travels at a faster rate in its shell than the inner electron, it is projected that Francium would be less reactive than Caesium. Similar to this, what is the element with the highest reactivity in the periodic table? When compared to other common metals such as iron and copper, which have no response when dropped into water, this is a significant difference. Cesium (caesium) is the metal that has been shown to be the most reactive in experiments.Īfter taking everything into consideration, which metal is the most reactive on the periodic table, and why?Īs the most reactive metal in the periodic table, caesium undergoes extraordinarily intense reactions, making it impossible to demonstrate in a classroom setting. This makes it simpler to remove the electron from the atom, and the atom becomes more reactive as a result. All alkali metals contain just one valence electron, which is the most common configuration. 
Francium is an alkali metal belonging to the first elemental group (IA).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
